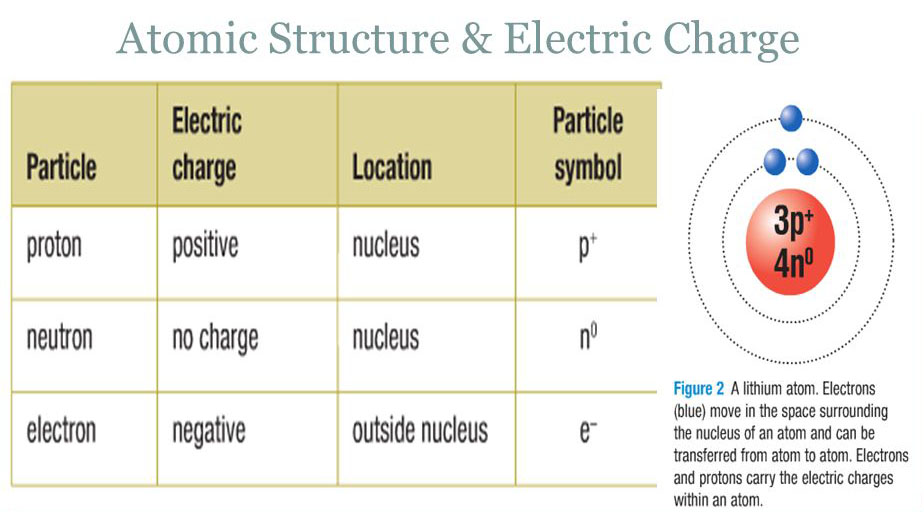
Objects that are neutral may be that way because of their inherent nature, like the neutron, but more commonly they are neutral because they contain equal numbers of negatively-charged particles (electrons) and positively charged ones (protons).Ĭharge is also a conserved quantity. Matter may be positively-charged, negatively-charged or neutral (not charged). We now know those to be negatively-charged electrons. In more controlled experiments, we can observe that charged objects exert invisible forces on one another.īenjamin Franklin, an early researcher in electricity and charge, assigned the label positive to the charges that tend to move the most. 
We understand charge because we can observe lightning, see sparking between wires and we can get an electric shock when things like fabrics rub together in the dry air of winter. MitoFit Preprint Arch 2020.4 doi:10.26124/mitofit:200004.Electric charge, like mass, is a fundamental property of matter. Gnaiger Erich (2020) Canonical reviewer's comments on: Bureau International des Poids et Mesures (2019) The International System of Units (SI) 9th ed.Gnaiger E (2020) Mitochondrial pathways and respiratory control.IUPAC Green Book 3rd Edition, 2nd Printing, IUPAC & RSC Publishing, Cambridge. Cohen ER, Cvitas T, Frey JG, Holmström B, Kuchitsu K, Marquardt R, Mills I, Pavese F, Quack M, Stohner J, Strauss HL, Takami M, Thor HL (2008) Quantities, Units and Symbols in Physical Chemistry.Bureau International des Poids et Mesures (2019) The International System of Units (SI).Keywords » charge Q el X » charge number z X » electrochemical constant f » elementary charge e » Faraday constant F » hydrogen ion versus proton » iconic symbols » motive entity » particle charge Q NXįundamental relationships » Avogadro constant N A » Boltzmann constant k » elementary charge e » Faraday constant F » gas constant R » electrochemical constant f SI and related concepts » International System of Units » elementary unit x » SI prefixes » International Union of Pure and Applied Chemistry, IUPAC » entity » quantity » dimension » format » motive unit » iconic symbols 
Therefore, the charge number of the hydrogen ion is z H + = Q NH +/ e = 1. The particle charge Q NH + per hydrogen ion is identical to the definition of the elementary charge e. To avoid too long and multiple subscript levels, Q NX is used instead of Q U X, and the ‘el’ is dropped from Q el N X. For consistency with this convention, the symbol Q elB or Q el X is used for indicating charge of a substance of type B or X, distinguished from particle charge as the quantity of charge per elementary entity X with symbol Q NX. This is opposite to the subscript in V B as the symbol for the volume of a substance of type B (e.g. The subscript in Q B indicates per elementary entity B. Ambiguity of Q B IUPAC ( Cohen 2008 IUPAC Green Book) defines the charge number as IUPAC: z B = Q B Therefore, electron charge (or proton charge) is clearly the charge per particle. Dividing ρ by the count concentration of electrons, we obtain the unit for the electron charge. For comparison, the name "charge density of electrons" is used by IUPAC with symbol ρ. The distinction of charge of particles versus charge per single particle is not made sufficiently clear by IUPAC, when defining "- e is the charge of an electron" - it must be corrected to "- e is the charge per electron". 2.1 Charge of the proton versus charge per protonĬommunicated by Gnaiger E () last update Ĭanonical comments on IUPAC definitions in the context of charge Charge of the proton versus charge per proton Proton charge is the elementary charge e, which is charge per count of protons.2 Canonical comments on IUPAC definitions in the context of charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
